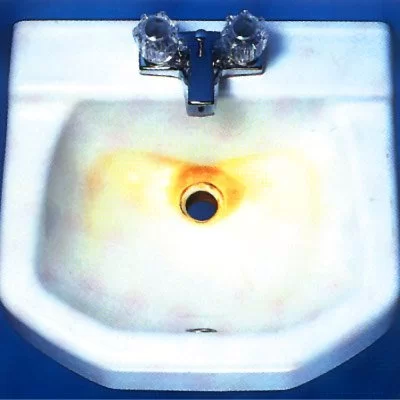
Rust or iron oxide develops when iron or iron elements in other materials begin to corrode as they react with moisture or water in the air or on the surface. Exposed metal surfaces and areas with high moisture content such as bathrooms are prone to rust development. The heavy application of cleaning products containing tri sodium phosphate may do the trick but works best when used in a paste form and left for a short time before removal with a damp cloth. Additionally, powders and mixtures containing oxalic acid compounds work well on stubborn or long-standing rust stains on porcelain, if used with a large amount of elbow grease. Again, creating a paste of crystals and hot water is the best way for people to utilize chemical compounds, while always taking appropriate precautions.
One alternative method is gentle scrubbing with a damp pumice stone directly on the rust stain. A homemade paste of hydrogen peroxide and cream of tartar, allowed to sit on the stain for a half hour before wiping with a damp cloth, might also do the trick. One green alternative to traditional chemical compounds includes natural, plant-based, nontoxic rust removers.
It is important to note that regular household bleach should never be viewed as a cleaning product for rust or rust stains. In fact, bleach sets the rust stain on the surface and makes it very difficult, if not impossible, to remove.